These Stem Cells are very beneficial for any health condition, whether it is a serious matter or only for staying strong and young; always providing them to patients with safe and effective therapies.
After a sperm fertilizes an egg, the latter divides into two cells, and then again, it divides itself in two and so on, becoming a hollow ball of cells called a Blastocyst; then, this process is interrupted for the harvest of PLURIPOTENT Stem Cells.
Optimally, PLURIPOTENT Stem Cells are harvested after the 4th or 5th day of divisions from the Blastocyst leftover from in vitro fertilization. The Blastocyst of 150 or so cells is then extracted and cultured in Petri dishes.
The 150 cells will divide into millions of ESCs, which have the capability of becoming any type of cell; these are known as Pluripotent. The cultured ESC’s are then mixed with plasma, this being the final step before infusion.
PLURIPOTENT Stem Cells are produced with several advanced scientific steps that transform skin cells into PLURIPOTENT Cells. No life is being produced or destroyed and there is only a transformation of old living cells into young and more effective ones.
PLURIPOTENT Stem Cells therapies give the knowledge and experience to make the processes safer and reliable. It’s time this method reaches every part of our world for those in need.
A pluripotent stem cell is an undifferentiated cell that has the potential to divide and differentiate into any of the 220 cell-types of the human body. Stem Cell Therapy (SCT), is the utilization of these cells in order to assist the body in the healing and regeneration of its existing cells.
Depending on the conditions, pluripotent stem cells can be delivered through the blood stream or directly to the organ that is in need of treatment. Within 30 minutes of their delivery, the body starts to repair its damaged organs and the healing process can be observed up to 10 days after the injection. Several injections might be needed depending on the severity of the conditions and the expected results.
Induced Pluripotent Stem Cells are prepared by introducing foreign genes into adult cells. These cells behave like PLURIPOTENT Stem Cells and are theoretically able to stimulate repair in the entire body. However, they are not considered fully PLURIPOTENT due to their foreign genes and this limits their range of application. Viruses are currently used to induce the reprogramming of adult cells, and this process must be carefully tested before they can lead to safe and effective treatment in humans because in animal studies, the virus used may sometimes cause cancers.
At Hino’s Medical Center we are developing powerful treatments for clinical trials that are changing the lives of our patients with heart, autoimmune, neurological, metabolic diseases, cancer and other illnesses.

Studies about the anti-aging of PLURIPOTENT Stem Cells on people between 50 and 70, and all of them described a significant improvement in their energy level.
The blood tests presented an increase of testosterone and estrogen in almost all of them as well as an increase in their telomere length by an average of 14% on the last months of the study. Therefore, that proves that with ESC treatments, patients present a reversion of their age at the DNA level, independently of their biological age.
PLURIPOTENT Stem Cells also help in the recovery from brain injury, making the brain’s repair mechanism to start over again. The transplanted Stem Cells create a bio-bridge that links the neural Stem Cell to the area of injury.
What are the differences between PLURIPOTENT Stem Cells, Adult and Umbilical Cord Stem Cells?
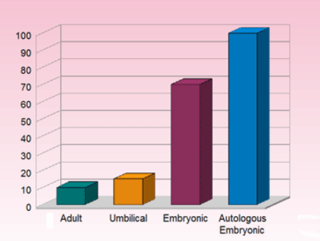
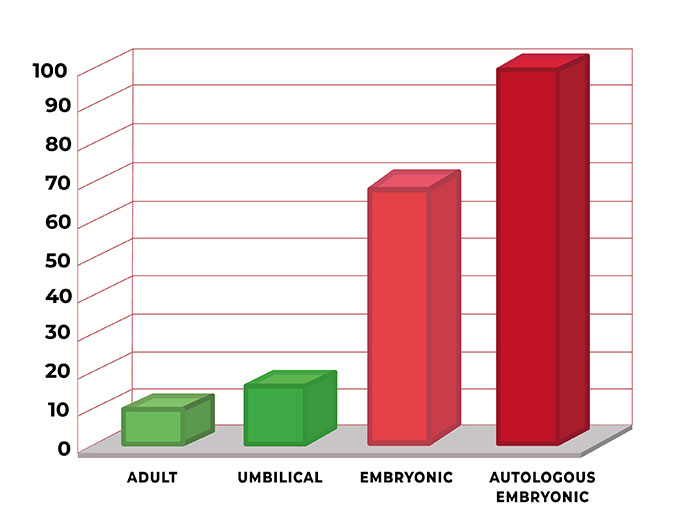
Adult Stem Cells are harvested from grown organs such as bone marrow, blood or fat tissue of the patient, and re-injected. Studies have concluded that adult stem cells are only able to develop into a limited number of cell types related to the tissue that the stem cells originally came from, in other words, they are multi-potent. Umbilical Stem Cells are adult stem cells taken from a baby’s Umbilical Cord. Risk of rejection is usually greater when using Umbilical cord stem cells. ESCs are the only pluripotent Stem Cells. They are also the only stem cells to produce anti-cancer proteins. Autologous cells have proven to be the best Stem Cells available, the ultimate healing and rejuvenating option, however the technology to produce them is time and labor extensive and requires highly skilled techniques.
Frequently Asked Questions
- Is it ethical to use PLURIPOTENT Stem Cells for therapy?
- How do HMC stem-cells differ from those offered by other providers?
- How does HMC ensure the quality of its stem-cells?
- Are there side effects and what are the risks, if any?
- I read that PLURIPOTENT stem-cells can cause cancer. Is it true?
- Where do the stem cells come from?
PLURIPOTENT stem cells are derived from a blastocyst which is the stage of an embryo 0-4 days after fusion of an egg and a spermatozoa.
A decade ago, in order to produce such a blastocyst and derive PLURIPOTENT stem cells from it, our scientists needed to retrieve eggs from a woman and collect sperm from a male and then fertilize the egg in a petri dish. Nowadays, it is no longer necessary to retrieve eggs or sperm to produce a blastocyst. Our scientists are able to produce an egg from a skin cell. They are also able to fuse such an egg with a fibroblast (skin cell) to produce an embryo.
In other words, our PLURIPOTENT stem cells are produced by transforming skin cells into PLURIPOTENT stem cells following several advanced scientific steps. The blastocyst produced in the process of this transformation is a transient stage where the skin cell is fully returned to its original PLURIPOTENT stage. No life is being produced or destroyed. There is only a transformation of old living cells into young and more effective ones.
HMC is a unique company in the world offering PLURIPOTENT stem cells. PLURIPOTENT stem-cells are up to 6x more potent in their ability to help the human body heal itself than that of adult-stem-cells or umbilical-stem-cells. While AUTOLOGUS PLURIPOTENT stem-cells are up to 10x more potent.
HMC stem cells are always delivered fresh from the laboratory culture. We never provide frozen stem cells to our partnered doctors as it would reduce the efficacy and number of stem-cells available for use by your body after injection.
We work with doctors to ensure that stem-cells are counted prior to injection, without any additional freezing. This ensures that patients always receive a minimum of 20 million stem-cells per injection. Some clinics or doctors purchase stem-cells in a frozen state, and simply ‘defrost’ them prior to injection, so even if there were 20 million stem-cells prior to freezing, by the time they reach your body, you are receiving less stem-cells than you were told, or possibly none at all!
Regardless of who you choose for your stem-cell therapy, make sure that the stem cell count is done in the last stage – prior to injection and without additional freezing.
To date, Stem Cell therapy has produced no negative side effects; nor are there any medically or scientifically anticipated risks outside that of a routine outpatient injection.
There is no scientific evidence that PLURIPOTENT stem cells may cause cancer. In fact, PLURIPOTENT stem-cells are the only stem-cells that have been scientifically proven to produce anti-tumor proteins that can shrink and even kill tumors. Others, including umbilical stem-cells and adult stem-cells, do not have this anti-cancer affect. In the many years in which our cells have been utilized, we have never seen such a problem after ESC treatment, on the contrary, we have seen tumors shrink and disappear.
Our PLURIPOTENT stem cells (ESC) are continually cultured in our labs. They were originally derived from a 100-200 cell blastocyst (either donated or cloned from a skin-cell) which is an early stage embryo, only 5 days old. At this very early stage, these cells have not yet become specialized in any way. In other words, they have not begun to transform (differentiate) into skin cells, bone cells, muscle cells etc.
Once a particular stem cell line has been established, it can be cultured for many years without losing potency or effectiveness and, most importantly, without the need of additional blastocysts.